Contact Us
University of California Cooperative Extension Ventura County
669 County Square Drive, Suite 100
Ventura, CA 93003
Phone: 805.645.1451
Fax: 805.645.1474
Office Hours:
Monday - Friday from 9 a.m. to 4 p.m.
The office will be closed for the following holidays:
Diseases of Palm Trees: Some New Some Old
Palm trees are increasing in popularity in Southern California landscapes. New construction and renovation of existing landscapes have increased the demand for several varieties of palms. Whether we like them or not palms are here to stay and their presence in the landscape can be quite formidable. The Canary island date palm can attain massive proportions; mature trees weighing several tons are not uncommon. Since many customers require “instant” landscapes, specimens of Canary Island date palm are often transplanted as mature trees into the landscape. Because some of these palms are so large the cost of their initial installation and maintenance can be quite high. Losses are expensive. Landscape managers, arborists and consultants are paying closer attention to palm diseases and problems because there is so much money involved with them. A single Canary Island date palm can represent a five to twenty thousand dollar investment.
Diseases of palms may be either biotic or abiotic. Biotic diseases are caused by pathogens and are usually fungal in etiology. Abiotic diseases are caused by re-occurring environmental conditions that compromise the long-term growth and development of the palm. Some palms show resistance to their biotic pathogens and to conditions which cause abiotic disease, but all palms are susceptible to mismanagement of water, the extremes of pruning and poor planting practices.
Diseases affect all parts of the palm tree; the roots, stem, leaves and fruit. For landscapes we are concerned primarily with diseases that harm the palm in such a way as to cause undesirable (not aesthetic) symptoms or predispose the palm to failure. Some palm diseases are deadly and threaten the survival of the infected/affected plant. Other diseases are cosmetic and never cause any long-term harm to the overall functioning of the plant.
Biotic Diseases
Palms are primarily attacked by fungi. Although bacterial diseases do occur on palms they are very rare if not absent in California landscapes. Most biotic diseases of palms are somewhat rare and occur on stressed or declining trees. Although we do not have a huge number of new palm diseases, we are seeing more and more diseases occur on palms as their popularity and planting densities increase. Sometimes the same pathogen Gliocladiium vermoeseni for instance can produce different diseases on different plants. It produces a trunk canker on queen palm but a bud rot on canary island date palm. Sometimes the pathogen is highly specific. Fusarium oxysporum f.sp. canariensis only produces the wilt disease in canary island date palm.
Root Rots
Phytophthora spp
Root rots in palms are most often caused by anaerobic soil conditions. There are however pathogens that can take advantage of slow growing palms. Phytophthora spp. has been cultured by many laboratories from palms in California, however; this pathogen is not reported in the literature for most of our common species. Undoubtedly the disease is occurring, but it just has not been adequately described yet for the species we grow here.
Diagnosis is made by culturing the fungus from the roots. The signs of infection can not be seen with a hand lens. Negative lab or culture results are a problem because it does not necessarily mean that the roots did not die from Phytophthora, only that the fungus was not recovered. Some times this disease is confused with the abiotic disease because the symptoms of rotted roots look the same.
Although many practitioners use metalaxyl (subdue) as a nostrum for root rot cure, its efficacy has not been well studied in California. Drying out the root ball, adjusting the irrigation, prevention of flooding are all helpful in slowing the disease.
Armillaria spp.
Armillaria causes root rot of many ornamental plants and has been reported on Phoenix canariensis in California and on Washingtonia robusta in Florida. I have seen Armillaria mellea on Rhaphidophyllum hystrix in Santa Barbara. Although Armillaria diseases of palm are not common, they should also not be taken for granted. We should not plant palms in a landscape where Armillaria is a known problem expecting complete resistance to the pathogen. Palms do get the disease and it really is not yet clear which palms if any, are the resistant ones.
Diamond Scale
Host Range and Occurrence
Diamond scale is a disease caused in Washingtonia filifera by Sphaerodothis neowashingtoniae Shear. The disease is only reported in California and is especially problematic near the coast or in valleys with coastal influence. In a recent field study of amendments for palm planting, I observed that Washingtonia palms growing in Long Beach were much more susceptible to Sphaeordothis than those planted in Irvine CA. Although we see the disease in Riverside, it is much less common on young trees. I have examined native palms in California desert oases (Borrego Springs and Twenty Nine Palms) and found no evidence of the disease. However, the pathogen should occur in the native range of W. filifera as it is its only host. As Washingtonias have been used to a greater degree in landscapes the occurrence of hybrids (W. filifera X W. robusta) are in greater abundance. These hybrids are somewhat susceptible to the disease, so that it is not uncommon to find occasional fruiting bodies on a palm that resembles a W. robusta. Washingtonia robusta that have not hybridized with W. filifera are completely resistant to diamond scale, while the native W. filifera are susceptible. There is some problem here for landscapers who wish to ensure the purity of their palms and plant resistant specimens. We have found that nurseries are not producing pure stock. Many of the palms sold today as W. robusta have some W. filifera in them. Because these palms are wind pollinated and both species are common to Southern California, the purity of seed sources is suspect. The only way to obtain genetically pure W. robusta plants may be to collect seeds from their native range in Mexico.
Symptoms signs and progression of the disease
The disease is called diamond scale because of the diamond shaped ascothecia or ascus bearing structures that form on the leaves of affected palms. Contrary to common belief, this is not a “scale” insect, but the fruiting body of the ascomycete fungus mentioned above. The diamond shaped structures hold the sexual spores of the fungus. The asexual stage of the fungus (anamorph) has not been identified. Since not all the spore stages of the fungus are know, an accurate life history of the organism is impossible to discern. There are many gaps in the knowledge of this fungus and its associated disease in Washingtonia. We do not know the native range of its occurrence, its time of year for infection, the life span of the spores, optimal temperature ranges for the fungus or all of the events that lead up to a successful penetration and infection of new hosts. What little is known has been from casual observation of the pathogen and its associated disease.
The earliest symptom after infection is a water soaked lesion on the frond of an infected plant. A hard black stroma begins to form soon after this—the beginning of the fruiting body. The stroma enlarges and continues to gain size over a period of weeks to months. As the stroma gains size, the leaf tissues around it begin to yellow and if on the midrib of the frond, elongate forming long yellow streaks. Yellowing symptoms around nearby infections tend to coalesce. By the time all of this is noticeable, the leaves are usually mature and in a relatively horizontal or downward pointing position on the palm. Newly emerged leaves are seemingly unaffected. New leaves probably do not stay in their upright position long enough for the disease to develop on them. By the time the fronds are downward pointing along the trunk they have yellowed or turned completely brown. Palms unaffected by the disease appear to have about two times as many green leaves as extensively infected palms.
The reduction of leaf area and the slowed growth associated with diamond scale is debilitating and leads to reduced vigor of Washingtonia filifera. While it is not believed that diamond scale kills Washingtonia, it certainly predisposes these palms to attack from the pink rot fungus that can cause its death.
Control of the disease
Here again we have a vacuum of knowledge. While thiophanate-meythl (Clearys 3336) has been used against diamond scale, we have no hard data on its efficacy, timing of application or re-treatment interval. The same holds for copper based fungicides. Fungicide efficacy trials on this pathogen are badly needed. Although pruning would eliminate some inoculum, it is not known if pruning away old fronds is helpful in controlling the disease because we do not know how long the spores live in old leaves. The best control of diamond scale is to avoid planting palms with W. filifera genetics in coastal landscapes. If you should happen to inherit some W. filifera with the disease, it will always be there for you. I know of no cases were it has been eradicated from living palms.

Figure 1. Early infection

Figure 2. Ascostroma forming

Figure 3. Mature ascostroma on rhacis

Figure 4. Frond death from diamond scale and stunting/death from pink rot
Pink Rot
Although we do not have funding to produce this newsletter in color we have made these issues available on the web in a color format to better highlight the figures. You can visit the Ventura County Cooperative Extension Web site at: http://ceventura.ucdavis.edu/. If you would like to have a color hardcopy they are available upon request 805-645-1458.
In 1923, Biourge named and described the pink rot fungus as a flesh, rose, or salmon-colored fungus which in Belgium had injurious effects on Areca palms grown in greenhouses. By 1924, Chevalier reported the same fungus to cause death to Archontophoenix cunninghamiana, Howea forsteriana, Washingtonia filifera and Washingtonia robusta. Much of this early history of the disease is summarized by Bliss, 1938. Discussions of the disease lay dormant for many years until Feather and others, 1989, described pink rot as part of a disease complex along with Fusarium oxysporum on Phoenix palms. There has been little or no work on the disease since Feather et al. published their research.
The pink rot disease of palms is perhaps the most ubiquitous palm disease in the landscape. It is caused by the fungus Gliocladium vermoeseni, (Pennicillium vermoeseni) a member of the fungi imperfecti. The perfect stage of the fungus is not known. The biology of the pink rot fungus is amazing in that it can produce billions of spores while growing on a single plant. Thus its spores are always present where palms are grown so there is no way to avoid it. The fungus is a weak pathogen in that it requires a wound or other plant stress factors that enable it to infect. Pink rot also preys on old specimens that are growing slowly and become susceptible due to reduced growth rates of the main bud. It appears to be associated with palms growing in humid coastal areas. Although it can occur in inland valleys, it is more prevalent along the coast. The host range of the fungus is large, covering several genera and species of palms.
The fungus causes an interesting variety of diseases. On Queen palms (Syagrus spp.) it causes a trunk rot (Fig. 4). Sometimes the spores of pink rot can be found under the pseudobark of the palm (Fig. 5). In King Palm (Archontophoenix cunninghamiana), spores of the fungus can almost always be found under the clasping leaf bases. When injured (by premature removal of the leaf bases or by pruning wounds) the fungus can cause a trunk rot and death of the tree (Fig 6).
Pink rot can be controlled preventatively by avoiding wounds that allow entry of the fungus. It also helps to avoid planting susceptible species (such as Washingtonia filifera) along the coast. For old or valuable specimens, fungicides can be applied to prevent pink rot infections. Thiophanate-methyl is often used for this purpose. Since T-methyl is weakly systemic, some applicators believe it to be helpful therapy on already infected plants. There has been little or no research on the effects of newer fungicides on pink rot control.
References
Bliss, D.E. 1938 The Penicillium Diseas of Ornamental Palms. Paper 393, UC Citrus Experiment Station, Riverside CA.
Biourge, P.H. 1923. Les moisissures du groupe Penicillium Link. Etude Monographique. (The molds of the group Penicillium Link. Monographic Study.) La Cellule 33 (1)7-331.
ChevalierA. 1924. Observations de NN. N. Patouillard et Poupion. Rev. Bot. Appl. 4:108-109.
Feather, T.V., Ohr H.D.,, Munnecke, D.E., and Carpenter, J.B. 1989. The occurrence of Fusarium oxysporum on Phoenix canariensis, a potential danger to date production in California.



Figure 6
Fusarium Wilt or NOT??
Fusarium wilt is one of the most destructive palm diseases in California landscapes. It results in the death of Phoenix canariensis, is incurable and easily spread by common pruning practices. Since P. canariensis palms are large and often require a crane to install, the cost of removal and replacement (with a non-susceptible species) is exorbitant. The disease is widespread, affecting palms in landscapes wherever P. canariensis is grown and the density of diseased individuals seems to be increasing. Since a single Canary Island Date palm can cost over $10,000, this disease is taken quite seriously. Recent articles have appeared in many Southern California newspapers (many of these with misinformation about the disease) and the disease was recently featured on many network television channels as well as the cable news network. Big palms mean big money and big losses when they get sick--and this is news!
The disease was first identified in California by Feather, Ohr and Munnecke (1979) and later further characterized by the same group of researchers (Feather et al. 1989). There has been no further substantive research on the problem in California. Feather et al. showed that the disease is transmitted mechanically by pruning equipment and that the pathogen (Fusarium oxysporum f.sp. canariensis) can infect and cause disease in the date palm. They also showed that the disease can occur in hot inland valleys and desert regions such as Anza Borego as well as in coastal landscapes. Feather et al. (1979) further defined a disease complex between Gliocladium vermoeseni and F. oxysporum occurring particularly in coastal landscapes where the pink rot pathogen (G. vermoeseni) is prevalent. The main outcome of Feather and others work was that we learned that the disease is both soil and pruning equipment borne, that it is fatal and that fungicides were ineffective. Little has changed since these conclusions were drawn. Trees continue to die and there is no effective therapy for diseased trees.
For managers of Canary Island date palms, angst is severe when contemplating the diagnosis of Fusarium wilt. For many years plant pathologists and consultants have coached clientele to look for one-sided frond death as a symptom of the disease. Fusarium infected trees typically display this symptom and it has been very diagnostic. However, in a recent consultation, I examined fronds of Canary Island Date palms displaying one sided wilt without the presence of Fusarium. It is essential that preliminary diagnoses are verified by isolation and identification of the fungus from infected plant material.
There are other shoot blights and stalk rots reported in the literature caused by Diplodia phoenicum (Farr et al., 1995). Also, infection of the base of the fronds by Gliolcadium vermoeseni can cause similar one-sided frond death and vascular discoloration. In my recent examination of one-sided frond death in Canary Island date palms I isolated Dothiorella spp. and observed the same fungus later fruiting on the samples. This has been a particularly abundant year for Dothiorella caused diseases and it comes as no surprise that this fungus is hosted by Phoenix.
Although infection by Dothiorella or Fusarium can produce identical one-sided death symptoms (fig. 1a,b), there are differences which are obvious on closer inspection. When slicing through the rachis of the frond, Fusarium infected samples will show browning in scattered vascular bundles—the overall look is a slight browning of the otherwise white tissue. Dothiorella causes a severe browning of all the cortical tissues including the xylem elements giving a broad band of dark tissue (figure 2). If the samples are retained in a moist cool place, they will often form fruiting bodies (pycnidia) that erupt through the epidermis of the rachis (figure 3). Microscopic examination of the fruiting bodies and spores will confirm the diagnosis.
These observations are of course preliminary in nature and I will be looking at a lot more palms in the near future as well as conducting experiments on pathogenicity of Dothiorella on Phoenix. The main point is that diagnosticians should be cautious about diagnosing Fusarium wilt solely on the basis of one-sided frond death symptoms and vascular discoloration.
References
Farr, D.F., Bills, G.F. Chamuris, G.P. and Rossman, A.Y. 1995. Fungi on plans and plant products in the United States. American Phytopathological Soc. St. Paul MN.
Feather, T.V., Ohr, H.D. and Munnecke, D.E. 1979. Wilt and dieback of canary island palm in California. California Agriculture 33:19-20.
Feather, T.V., Ohr, H.D., Munnecke, D.E. and Carpenter, J.B. 1989. The occurrence of Fusarium oxysporum on Phoenix canariensis, a potential danger to date production in California.

Figure 1a Dothiorella

Figure 1b Fusarium Wilt

Figure 2. Vascular browning of Dothiorella infection

Figure 3. Pycnidia of Dothiorella
Abiotic Diseases of Palms
As with other kinds of plants the abiotic diseases are perhaps more common than the biotic ones. This is largely due to mistakes in the planting, maintenance or treatment of palms in landscapes. Abiotic diseases result from an extremes of water, light, temperature or the chemical environment surrounding the roots of palms.
The trouble is the greatest in large transplanted specimens where the new planting site is highly modified from its original state and the palm is planted so that there is an excess of soil (sometimes several feet) over the rootball. When this soil change involves sand and organic matter placed over a heavier clay or loam soil the result is an anaerobic breakdown of the amendment, release of hydrogen cyanide and other gases and the death of palm roots and eventually death of the tree. See figure below.
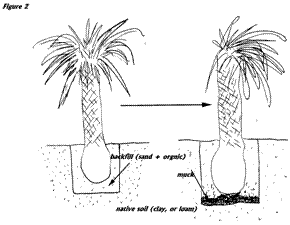
Pruning is another practice that can lead to the death of palms through depletion of carbohydrates in the stem of the palm. As more and more leaves are removed the palm runs out of photosynthetic machinery to make its food, stored starch reserves are used up and the palm eventually dies.

The palms pictured above are transplants. All have been excessively pruned and the one on the far left is probably not coming back. As long as fronds are not brown, they contribute photosynthate to the tree and should remain. Over pruning palms causes them to lose vigor and decline.
When palms are grown closely they etiolate just like other monocots and produce long narrow stems. These trees when planted out can often break due to lack of the forest that once protected them and lack of the strength in their fronds necessary to resist wind. See the queen palm in the photo below.

Palms like all plants thrive in environments that they are capable of growing in. When changes are made that are drastic such as dramatic changes in light, moisture status, salinity, or temperature, they will express symptoms of damage. These symptoms may also suggest that the palms are now predisposed to attack from the biotic pathogens mentioned above and that a new disease may be on the horizon for that individual. We often see palms with pink rot after a cold winter and freezing temperatures. Good disease management requires an understanding of the proper growing conditions for the palms and the potential for disease from the pathogens that can attack it. Creating conditions favorable for palm growth but not pathogen proliferation will be helpful.
Labs that Analyze Palm Tree Samples
This list is not meant to be an endorsement of the following firms. These are labs known to me that conduct palm tree pathology analyses. Other labs may exist and may have equal capabilities. No endorsement of these labs is intended nor is any quality statement being made about labs listed here or not listed.
1.) Soil and Plant Lab, Santa Anna, CA
2.) Dr. Don, Ferrin, Riverside, CA
3.) Richard Cullen (for Fursarium oxysporium f.sp. canariensis id)
Florida Extension Plant Disease Clinic
University of Florida
Institute of Food and Agricultural Science
Building 78 Mowry Rd.
PO BOX 110830
Gainesville, FL 32611-0830
352-392-1795
